 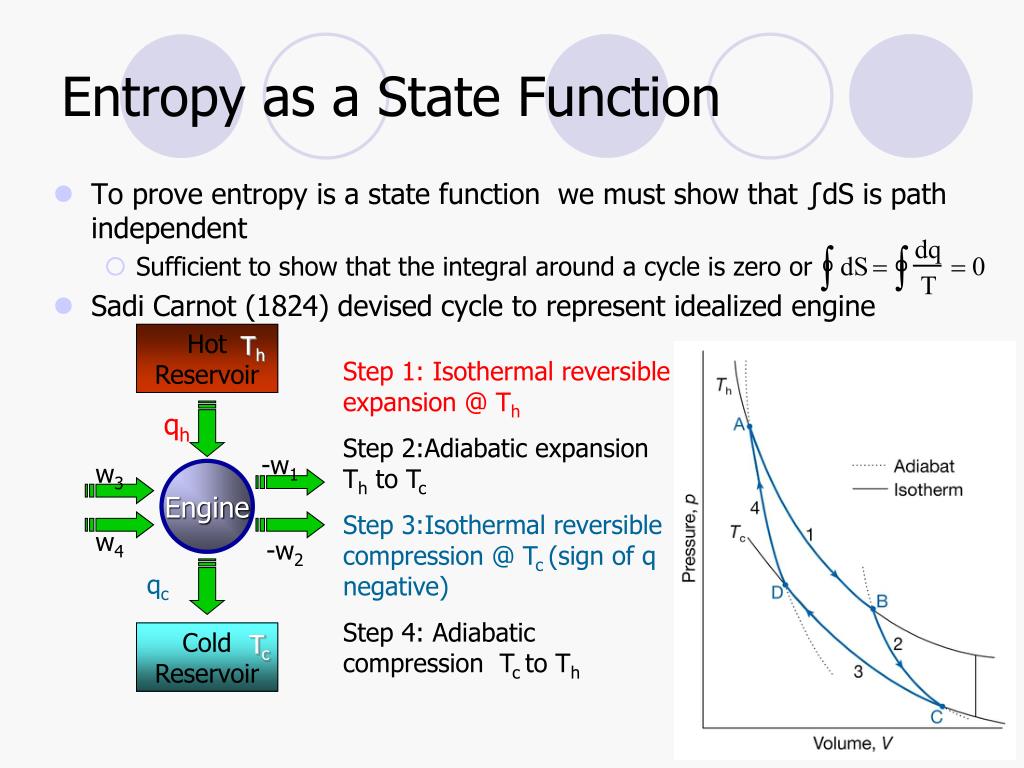
S(N V E) is a material speci c equation of state from which all thermodynamic relations of this material can be derived. An irreversible process increases the total entropy of the system and its surroundings. 3 Boltzmann’s entropy expression S V N Eare all state functions in thermodynamics. However, the impossibility occurs in restoring the environment to its own initial conditions. 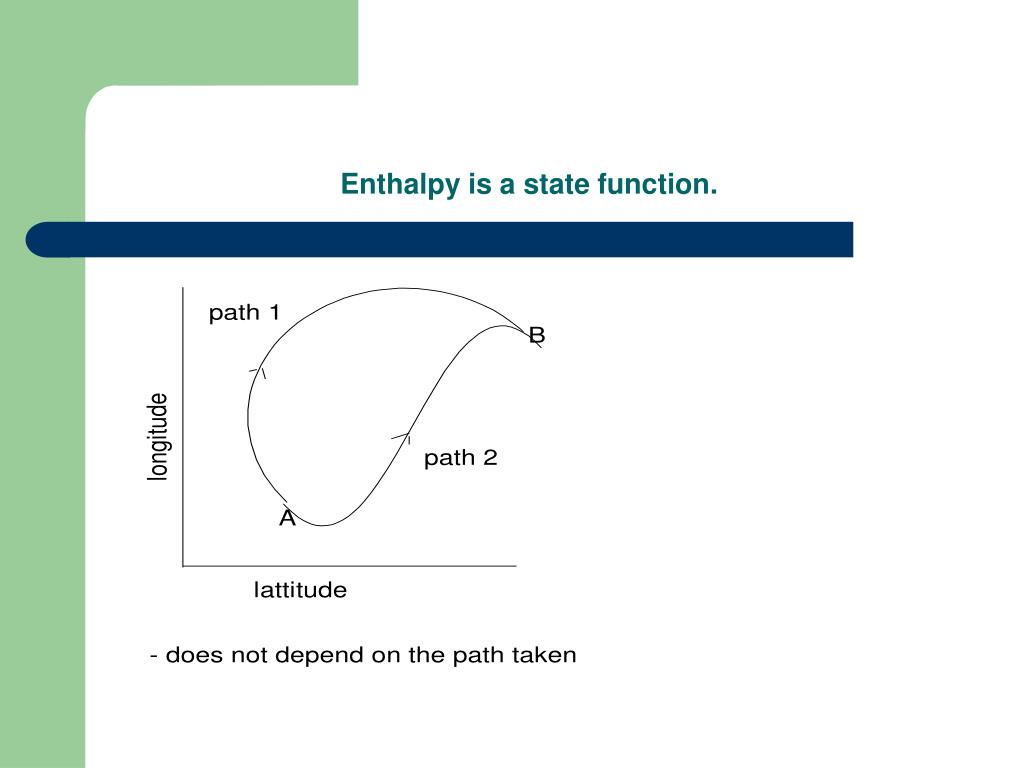
The second law of thermodynamics was previously described with the Kelvin-Planck and Clausius statements. Because entropy is a state function, the change in entropy of the system is the same whether the process is reversible or irreversible. 6.5.4 T he second law of thermodynamics e xpressed in terms of entropy generation, S gen \displaystyle\oint\dfrac is the change of entropy in the subsystem, i, in the universe. The inequality of Clausius states that for any cycle, reversible or irreversible, there exists the following relation: The concept of entropy provides deep insight into the direction of spontaneous. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.  
Entropy and the Second Law of ThermodynamicsĦ.5 Entropy and entropy generation 6.5.1 The inequality of Clausius The most important conclusion of this definition is that entropy, being a state function, is not a function of time. entropy, the measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work. The relationship between entropy, as a non-state function. A system's entropy plus the entropy of its surroundings will be larger than zero.6. Entropy is discussed as a fundamental goal function in the far from equilibrium framework. The change in total entropy is always positive. Thermodynamically, complete heat transmission into work is not possible without wasting a certain amount of energy. The second law of thermodynamics is best expressed in terms of a change in the thermodynamic variable known as entropy, which is represented by the symbol S.Entropy, like internal energy, is a state function. Since it is a state function, the change in entropy will only depend on the initial and. Note: Thermodynamically, all naturally occurring spontaneous reactions are irreversible. Entropy measures the degree of disorder or randomness of a system. Thermodynamically, all naturally occurring spontaneous reactions are irreversible. Entropy is a state function since it depends not only on the start and end states, but also on the entropy change between two states, which is integrating tiny entropy change along a reversible route. More specifically, the second law of thermodynamics states that as one goes forward in time, the net entropy (degree of. The First Law of Thermodynamics states that. It is obvious from these instances that when regularity decreases, entropy increases.Įntropy is a state function since it depends not only on the start and end states, but also on the entropy change between two states, which is integrating tiny entropy change along a reversible route. 3rd Law of Thermodynamics - A perfect crystal at zero Kelvin has zero entropy. A system with more unpredictability has a higher temperature than one with a lower temperature. Entropy increases when reactants break down into a larger number of products during chemical processes. Note that in an isolated system, the greater the disorder, the higher the entropy. Entropy is a scalable property, meaning it grows in proportion to the size or scope of a system. It is denoted by the letter S, although in the normal state, it is denoted by the letter\. It is determined by the condition of the system rather than the path taken. The change in entropy (delta S) is equal to the heat transfer (delta Q) divided. It has a wide range of applications in chemistry and physics, biological systems and their relationships to life, cosmology, economics, sociology, weather science, climate change, and information systems, including telecommunications. The second law states that there exists a useful state variable called entropy. The word and idea are utilised in a wide range of areas, from classical thermodynamics, where it was originally identified, through statistical physics' microscopic description of nature, to information theory's principles. A state function specifies a system's equilibrium state, as well as the kind of system.Įntropy is a quantifiable physical characteristic that is most frequently linked with a condition of disorder, unpredictability, or uncertainty. gas, liquid, solid, crystal, or emulsion), not on the path it took to get there. Hint: A state function, also known as a function of state or a point function, is a function defined for a system that connects several state variables or state quantities and is dependent only on the system's current equilibrium thermodynamic state (e.g. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
